 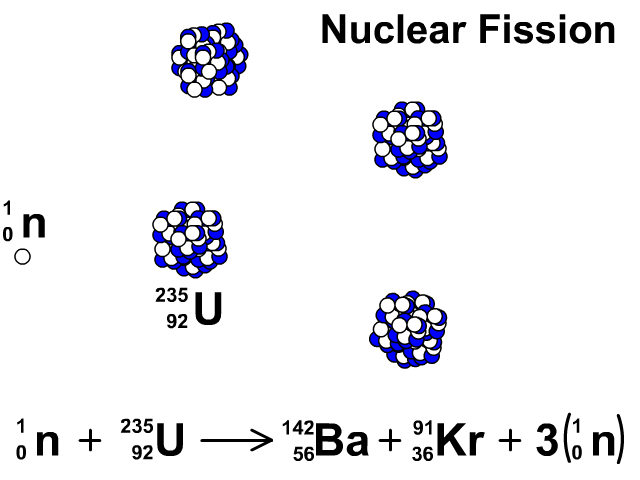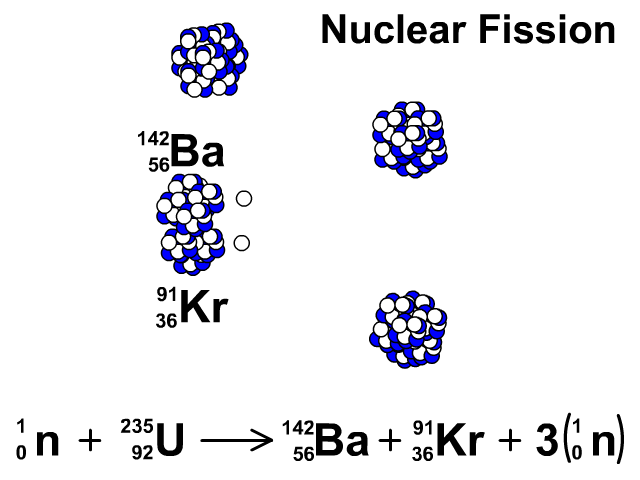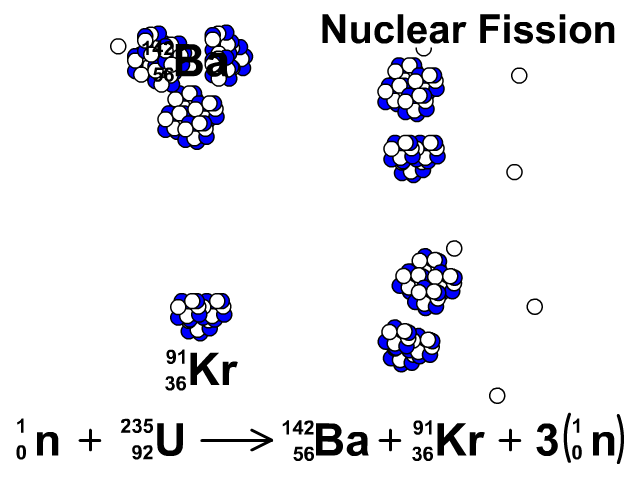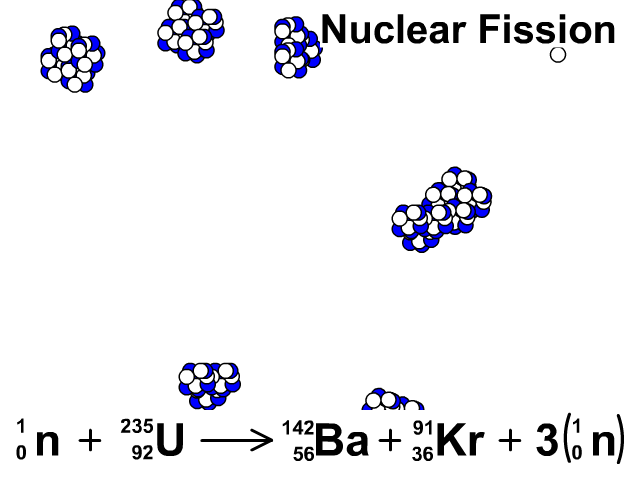
Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them.\nonumber \]Īgain, we see that the total number of mass units and of protons is equal on both sides of the equation. Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". (b) Show that the fission of 1 kg of uranium-235 releases approximately a million times more energy than the combustion of. In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. 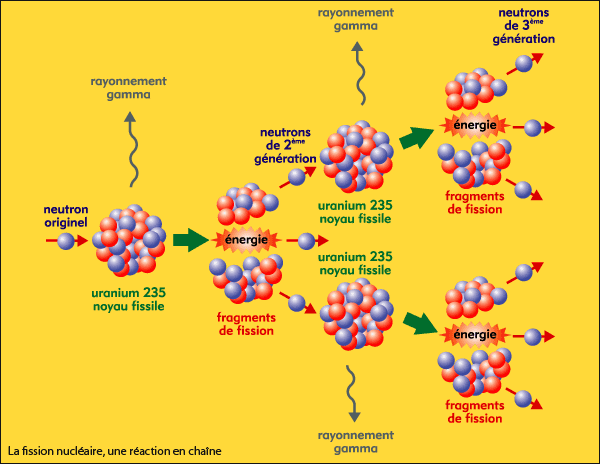
As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. To put the reactor into operation, the control blades are raised very slowly. When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Question: 3 (d) Consider the fission of uranium-235 in accordance with the nuclear equation: 235 1 141 x U + n + Ba + Kr +3 n 92 0 y 36 0 371 i) What are. Boron has the property of absorbing neutrons without re-emitting any. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. Although fairly simple in theory, an atomic bomb is difficult to produce, in part because uranium-235, the isotope that undergoes fission, makes up only 0. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. The MIT Research Reactor is used primarily for the production of neutrons. It was later determined that of the three isotopes of naturally occurring uranium, only uranium-235 undergoes nuclear fission (Equation 4). (b) The diagram shows the nuclear fission process for an atom of uranium-235. (LWR) can start with 3 235U + 97 238U (a mixture enriched with respect to natural uranium). Hence, the possibility exists for creating a chain reaction. A neutron is absorbed by a uranium-235 nucleus, turning it briefly into an excited uranium-236 nucleus, with the excitation energy. (b) The following nuclear equation represents the fission of uranium-235 (U-235). Each time a U-235 nucleus splits, it releases two or three neutrons. This process is known as fission (see diagram below). When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. The reaction requires the input of a neutron and involves the. Describe nuclear fission and write a nuclear equation to describe the fission of U-235 Uranium-235 undergoes neutron-Induced fission to produce Xe-144 and Sr-90. 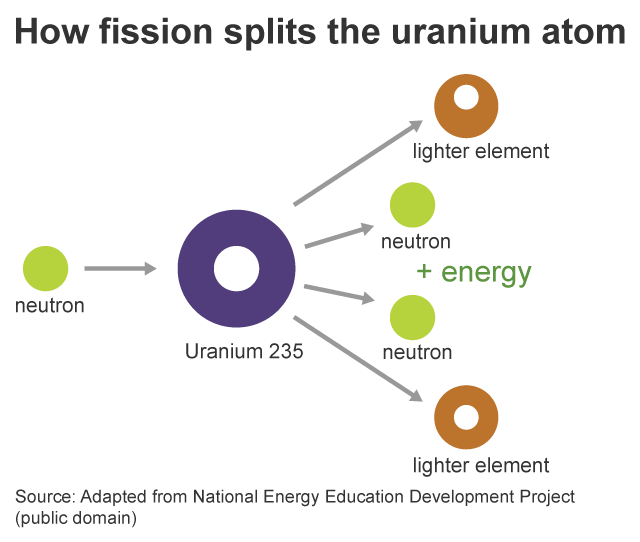
The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. The fission reaction of uranium-235 results in the formation of barium-140 and krypton-93. For fast neutrons, its fission cross-section is on the order of barns. In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235. Uranium 235 is a fissile isotope, and its fission cross-section for thermal neutrons is about 585 barns (for 0.0253 eV neutron). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
